Project mission
EXOTrainer
addresses the introduction of wearable gait exoskeletons for the therapy of
children affected by Spinal Muscular Atrophy (SMA). Although it is a rare
disease, SMA is the first cause of child mortality, affecting children of both
gender, of all races, in all countries. There is no cure for SMA, and treatment
focuses on maintaining the physical state of the patient, and attempting to
delay the onset of scoliosis. Walking is key for this purpose and EXOTrainer
provides a better therapy approach through wearable gait exoskeletons.
EXOTrainer builds over available technology and addresses a new target group
and different diseases, as current commercial devices are targeted to adult paraplegics.
EXOTrainer targets children improving the therapy of SMA and potentially other
rare diseases. EXOTrainer will have an impact on children’s quality of life and
life expectancy. The new therapy can potentially reduce the welfare costs to
families in 25.000€ per year and per family and even reduce the cost to the
healthcare systems by an improved and personalized therapy.
NOVELITY
EXOTrainer will make progress on the technical problems of commercial exoskeletons for SMA children.
EXPECTED IMPACT
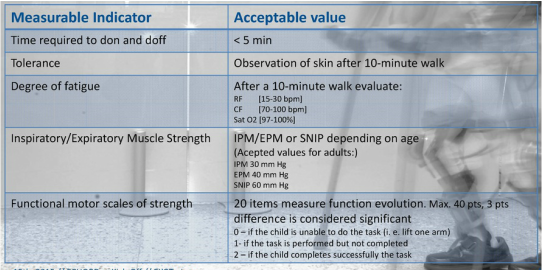
The EXOTrainer experiment ends with a clinical proof of concept. The main objective of this clinical trial is to demonstrate the feasibility, safety and tolerance of the device, i.e. the child with SMA II has never walked, and will be able to walk with the exoskeleton without side effects. Therefore the first impact of EXOTrainer is providing the ability to walk. After that, the clinical protocol includes a series of measurable parameters that will be translated into quantitative measurements of impact on feasibility and tolerance, which include:
|
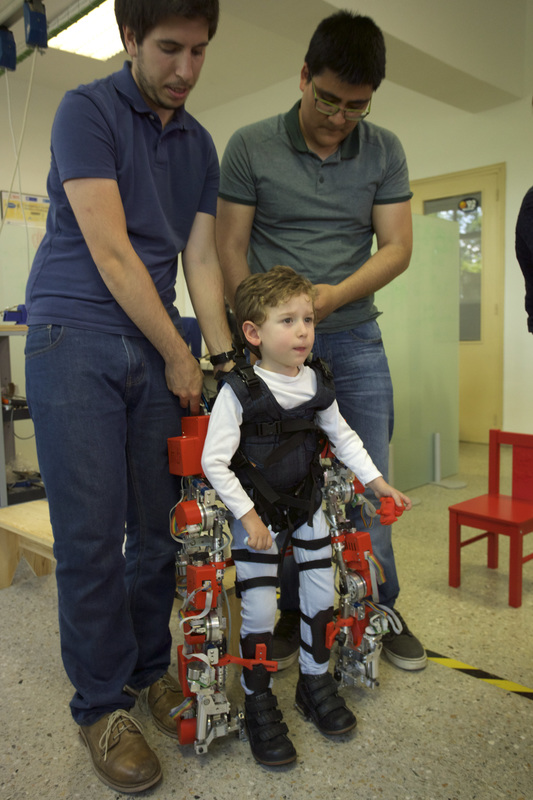
Conceptual sketch of EXOTrainer exoskeleton for gait training with attached frame.
Pediatric exoskeleton for gait training of SMA children
|
Echord++ EXOTrainer